-
What’s patent eligible and what’s not: patent eligible subject matter comparison – China

As a national strategy, China is trying to move from being a world manufacturing factory to being an innovation leader. Since the introduction of the “Law of the People’s Republic of China on Progress of Science and Technology” in 2008, China has made staggering progress toward promoting the scientific and technological innovations, which is evidenced by…
-
Summary of the America Invents Act

Repost from AIPLA: http://www.aipla.org/advocacy/congress/aia/Pages/summary.aspx Filing Reforms Examination Reforms Third Party Reforms Additional Key Reforms Other Provisions Thanks for reading. Contact us at info@epimedresearch.com
-
Maximizing exclusivity for pharmaceutical products

1. The Hatch-Waxman Act The Drug Price Competition and Patent Term Restoration Act, commonly known as the “Hatch-Waxman” Act, was enacted in USA in 1984. The Act aims to strike a balance between the drug innovators and generic manufacturers by permitting limited extensions of patent term to compensate for market time lost during the drug approval…
-
On-sale bar to patentability could be triggered by an order to your own OME manufacturer

On-sale bar is a patentability bar derived from 35 U.S.C. 102 of the U.S. Patent Law. Under 35 U.S.C. 102(a), a person shall be entitled to a patent unless- (1) the claimed invention was patented, described in a printed publication, or in public use, on sale, or otherwise available to the public before the effective filing…
-
Patent Eligibility of Gene: isolated gene not patent eligible in US
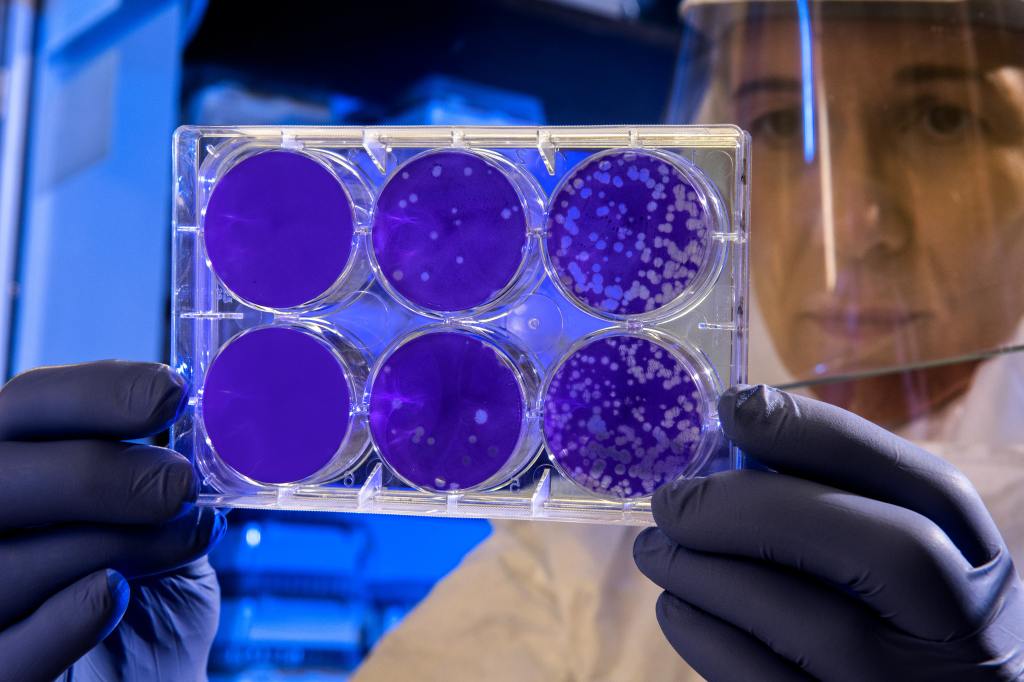
Well, it’s final—isolated gene is no longer patent eligible in US. The multi-years long, contentious and emotional fight on the gene patentability climaxed with a straightforward decision from U.S. Supreme Court in Association for Molecular Pathology v. Myriad, 569 U.S.____ on June 13, 2013. The case began with Myriad’s identification and sequencing of the two breast cancer-causing…
-
Climbing up to the World Stage: the Third Amendment to the Chinese Patent Law

In a showing of its desire to transform from a world’s factory into an innovation-oriented country, China recently announced the enactment of the third amendment to the Chinese Patent Law. Since adopting its original Patent Law in 1984, China has enacted two amendments, the first in 1992 and the second in 2000. The first amendment…
-
Summary and Implication of Phillips v. AWH Corp.

Introduction On July 12, 2005 , the Federal Circuit issued its en banc decision in Phillips v. AWH Corp., addressing fundamental questions regarding patent claim construction. See Phillips v. AWH Corp., No. 03-1269, -1286 (Fed. Cir. Jul. 12, 2005 ). The decision clarifies a handful of issues central to the claim construction process, in particular, the emphasis that is to be…
